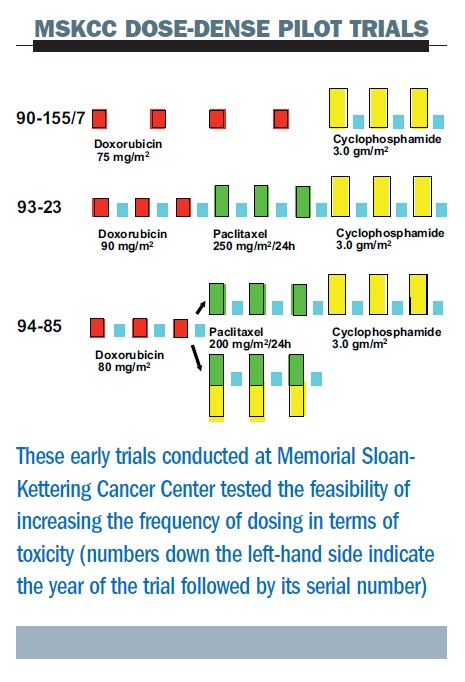
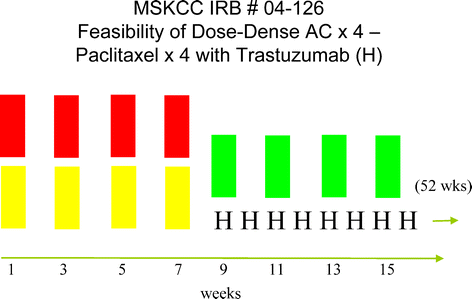
The safety of dose-dense doxorubicin and cyclophosphamide followed by paclitaxel with trastuzumab in HER-2/neu overexpressed/amplified breast cancer. | Semantic Scholar

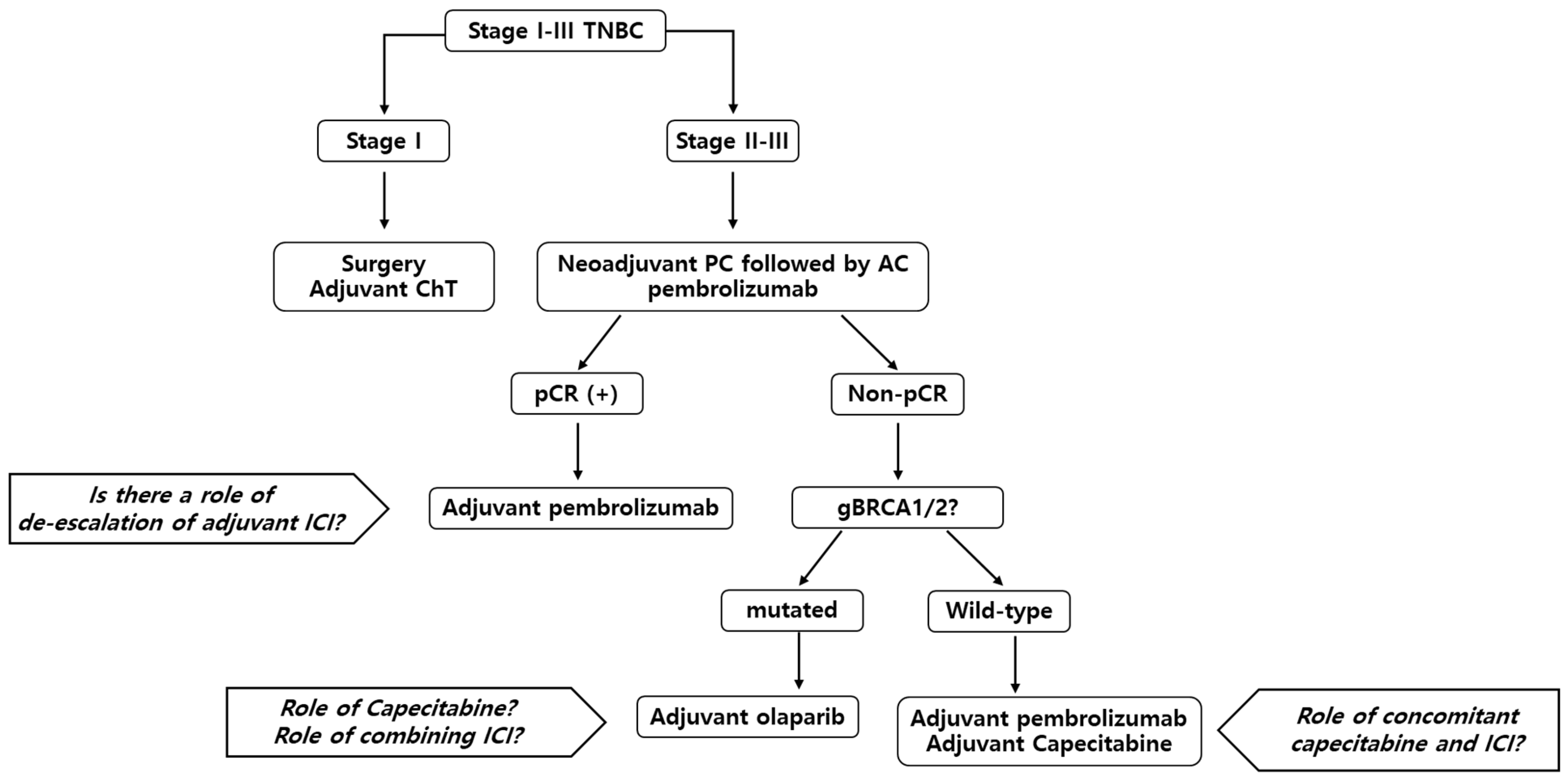
Neoadjuvant durvalumab plus weekly nab-paclitaxel and dose-dense doxorubicin/cyclophosphamide in triple-negative breast cancer | npj Breast Cancer

Phase 2 Study of Dose-Dense Doxorubicin and Cyclophosphamide Followed by Eribulin Mesylate With or Without Prophylactic Growth Factor for Adjuvant Treatment of Early-Stage Human Epidermal Growth Factor Receptor 2–Negative Breast Cancer -

Neoadjuvant MEDI4736 Concomitant With Weekly Nab-paclitaxel and Dose-dense AC for Stage I-III Triple

Timing of AC chemotherapy, dexamethasone dosing, and PCP diagnosis on a... | Download Scientific Diagram
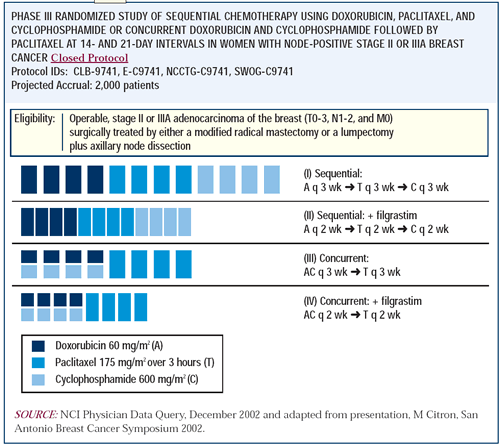
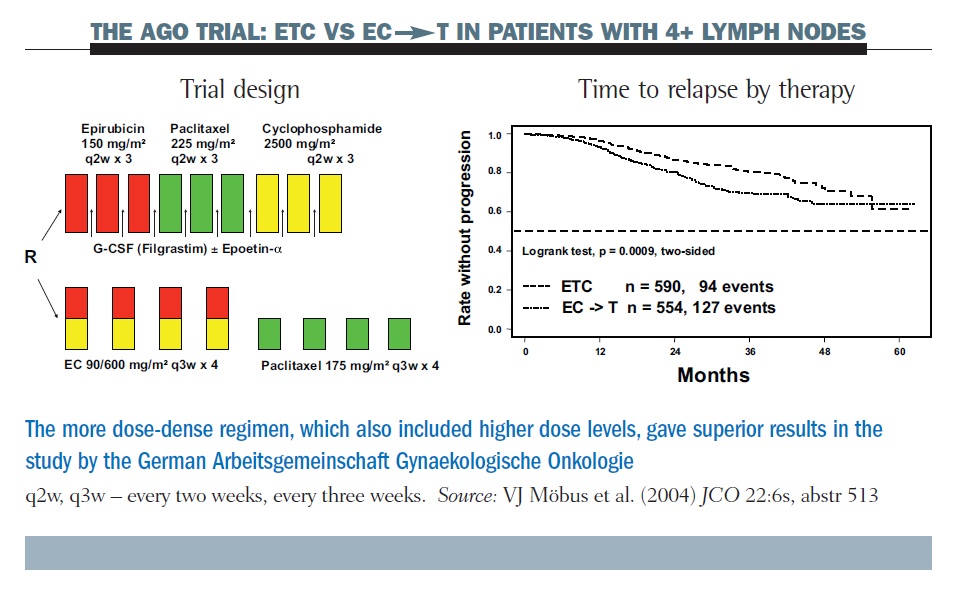
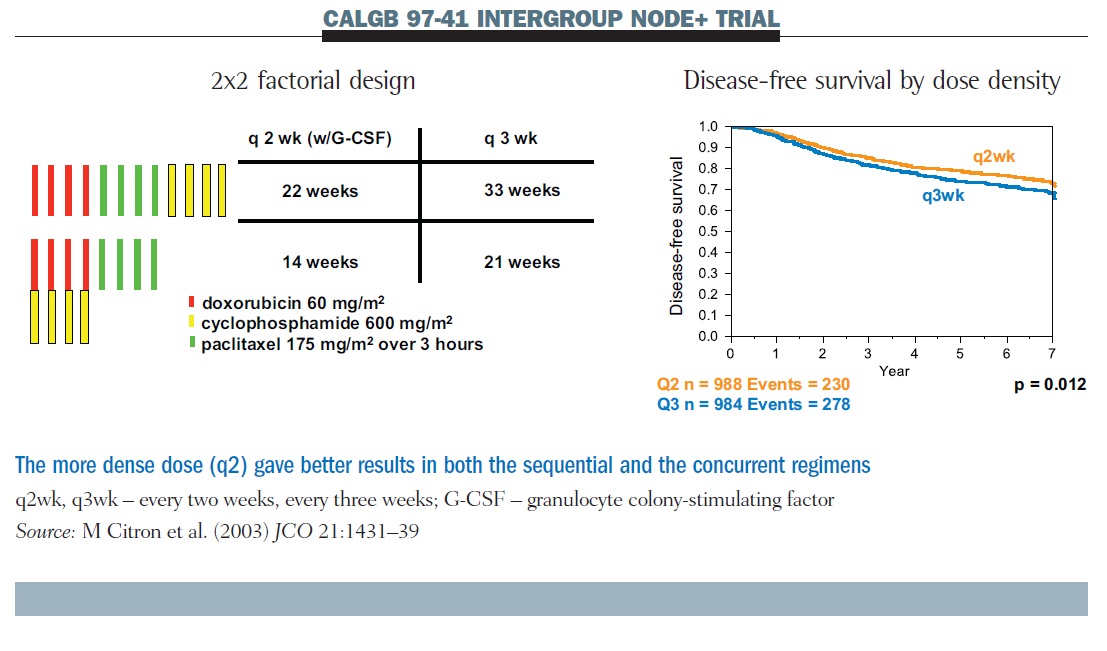
Randomized Trial of Dose-Dense Versus Conventionally Scheduled and Sequential Versus Concurrent Combination Chemotherapy as Post
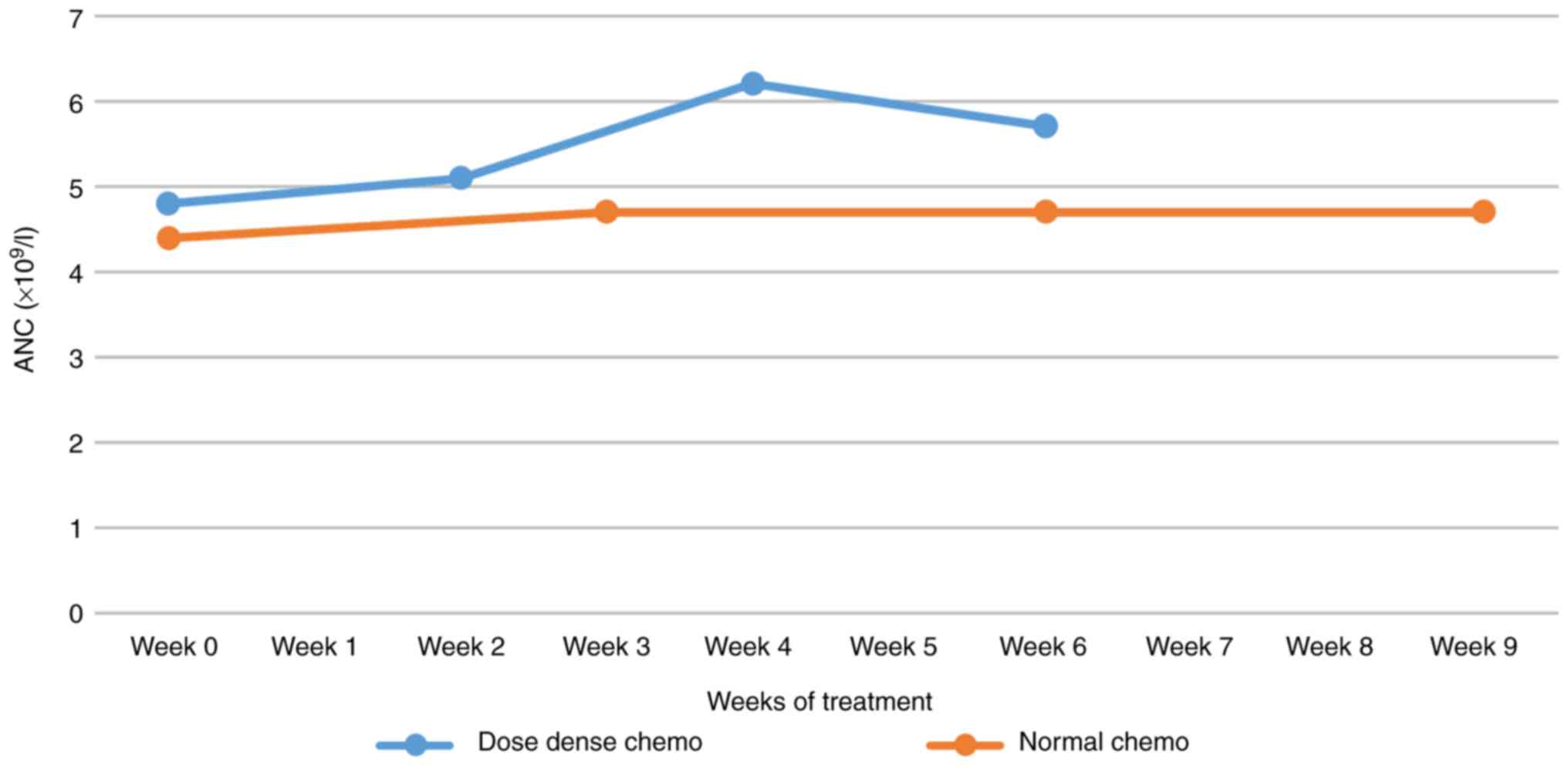
Average Hb evolution in dose-dense vs. normal AC chemo. Hb, hemoglobin. | Download Scientific Diagram

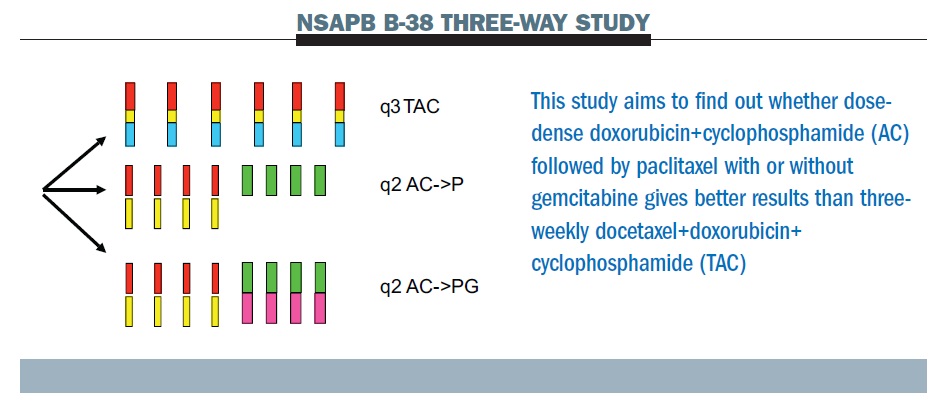
Fluorouracil and dose-dense adjuvant chemotherapy in patients with early-stage breast cancer (GIM2): end-of-study results from a randomised, phase 3 trial - The Lancet Oncology
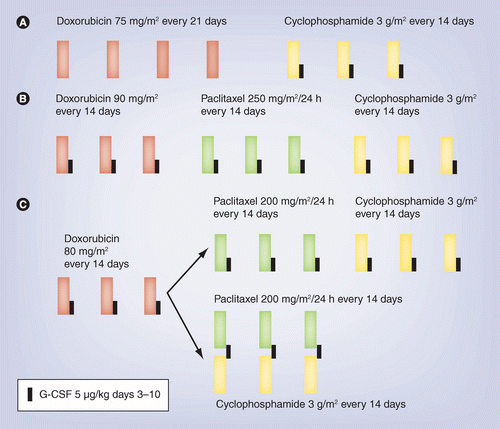
![PDF] A Feasibility Study of Bevacizumab plus Dose-Dense Doxorubicin–Cyclophosphamide (AC) Followed by Nanoparticle Albumin–Bound Paclitaxel in Early-Stage Breast Cancer | Semantic Scholar PDF] A Feasibility Study of Bevacizumab plus Dose-Dense Doxorubicin–Cyclophosphamide (AC) Followed by Nanoparticle Albumin–Bound Paclitaxel in Early-Stage Breast Cancer | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/3394304ab7e935738281c7fd73f48d28332ddc37/3-Figure1-1.png)
PDF] A Feasibility Study of Bevacizumab plus Dose-Dense Doxorubicin–Cyclophosphamide (AC) Followed by Nanoparticle Albumin–Bound Paclitaxel in Early-Stage Breast Cancer | Semantic Scholar

Weekly dose-dense chemotherapy in first-line epithelial ovarian, fallopian tube, or primary peritoneal carcinoma treatment (ICON8): primary progression free survival analysis results from a GCIG phase 3 randomised controlled trial - The Lancet
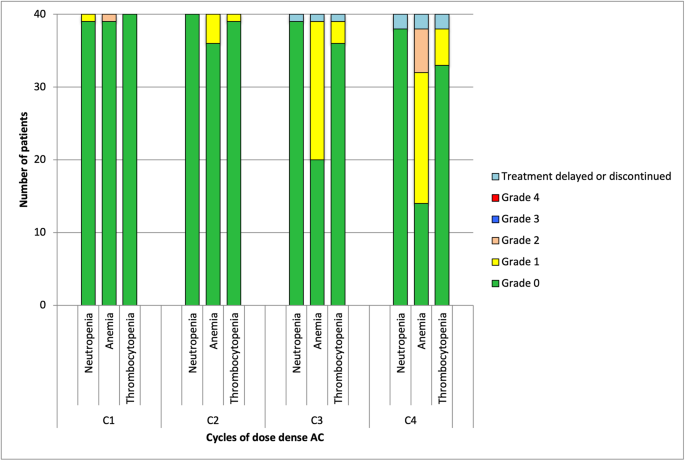
An observational study of dose dense chemotherapy with lipegfilgrastim support in early breast cancer | BMC Cancer | Full Text

SWOG S 0800 ( NCI CDR 0000636131 ) : addition of bevacizumab to neoadjuvant nab-paclitaxel with dose-dense doxorubicin and cyclophosphamide improves pathologic complete response ( pCR ) rates in inflammatory or locally