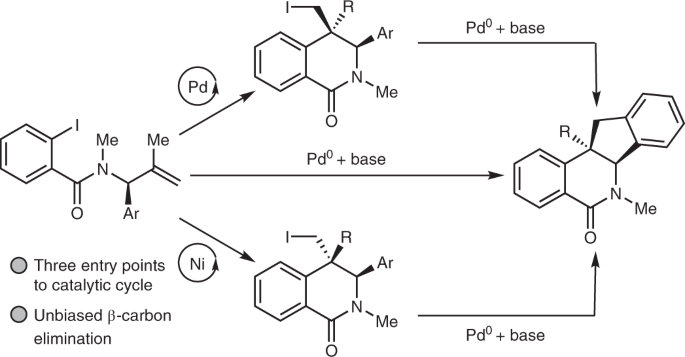
Palladium‐Catalyzed C–S Bond Formation: Rate and Mechanism of the Coupling of Aryl or Vinyl Halides with a Thiol Derived from a Cysteine - Moreau - 2005 - European Journal of Organic Chemistry -

Formation of Palladium(0) Complexes from Pd(OAc)2 and a Bidentate Phosphine Ligand (dppp) and Their Reactivity in Oxidative Addition | Organometallics

PDF) Palladium-mediated intramolecular C–N bond formation involving allyl substituted pyridines. Application to a novel strategy for the synthesis of the skeleton of berberinium derivatives | Mike Robitzer, Michel Pfeffer, and Gerard Van

Formation of Palladium(0) Complexes from Pd(OAc)2 and a Bidentate Phosphine Ligand (dppp) and Their Reactivity in Oxidative Addition | Organometallics

Formation of XPhos‐Ligated Palladium(0) Complexes and Reactivity in Oxidative Additions - Wagschal - 2019 - Chemistry – A European Journal - Wiley Online Library

Formation and structures of palladium-cyanomethyl complexes generated under unexpectedly mild conditions: quantifying the acidification of acetonitrile coordinated to a palladium center | Inorganic Chemistry | ChemRxiv | Cambridge Open Engage

Unexpected Formation of Hexasubstituted Arenes Through a Twofold Palladium-Mediated Ligand Arylation | Organic Chemistry | ChemRxiv | Cambridge Open Engage

Formation of palladium complexes of ligands 2a, 5 and 7. (A) Formation... | Download Scientific Diagram

Homogeneous Palladium Catalyst Suppressing Pd Black Formation in Air Oxidation of Alcohols | Journal of the American Chemical Society

Palladium-Catalyzed Direct Intramolecular C-N Bond Formation: Access to Multisubstituted Dihydropyrroles
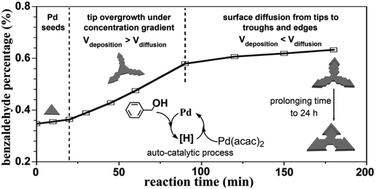
Formation of palladium concave nanocrystals via auto-catalytic tip overgrowth by interplay of reduction kinetics, concentration gradient and surface diffusion - Nanoscale (RSC Publishing)

Homogeneous Palladium Catalyst Suppressing Pd Black Formation in Air Oxidation of Alcohols | Journal of the American Chemical Society
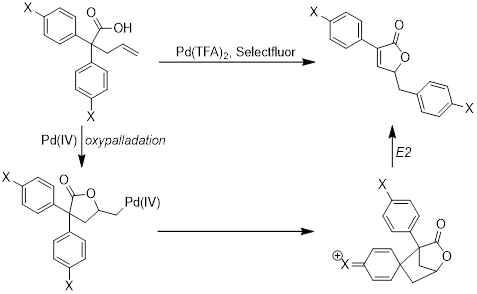
Report: Development of a Palladium-Catalyzed Oxidative Aryl Transfer Reaction (62nd Annual Report on Research Under Sponsorship of The American Chemical Society Petroleum Research Fund)
![PDF] Palladium-catalyzed C-C bond formation of arylhydrazines with olefins via carbon-nitrogen bond cleavage. | Semantic Scholar PDF] Palladium-catalyzed C-C bond formation of arylhydrazines with olefins via carbon-nitrogen bond cleavage. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/43c3b24a29c631438999caf7ba3027f907f307ae/17-Figure1-1.png)
PDF] Palladium-catalyzed C-C bond formation of arylhydrazines with olefins via carbon-nitrogen bond cleavage. | Semantic Scholar

Palladium-Catalyzed C–P Bond-Forming Reactions of Aryl Nonaflates Accelerated by Iodide - ScienceDirect
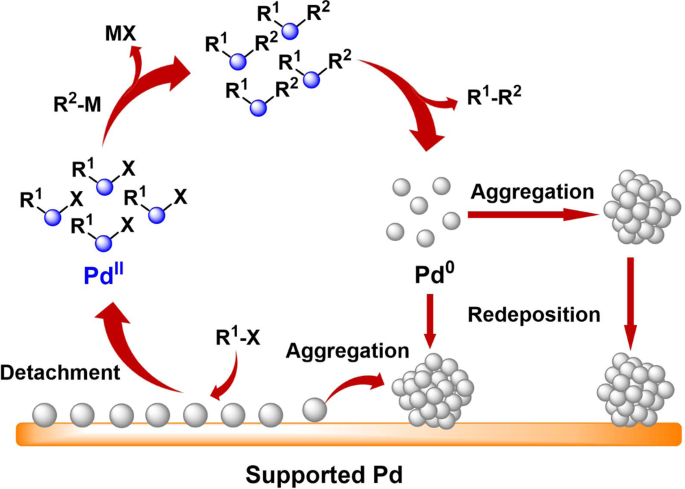
Enhancing stability by trapping palladium inside N-heterocyclic carbene-functionalized hypercrosslinked polymers for heterogeneous C-C bond formations | Nature Communications
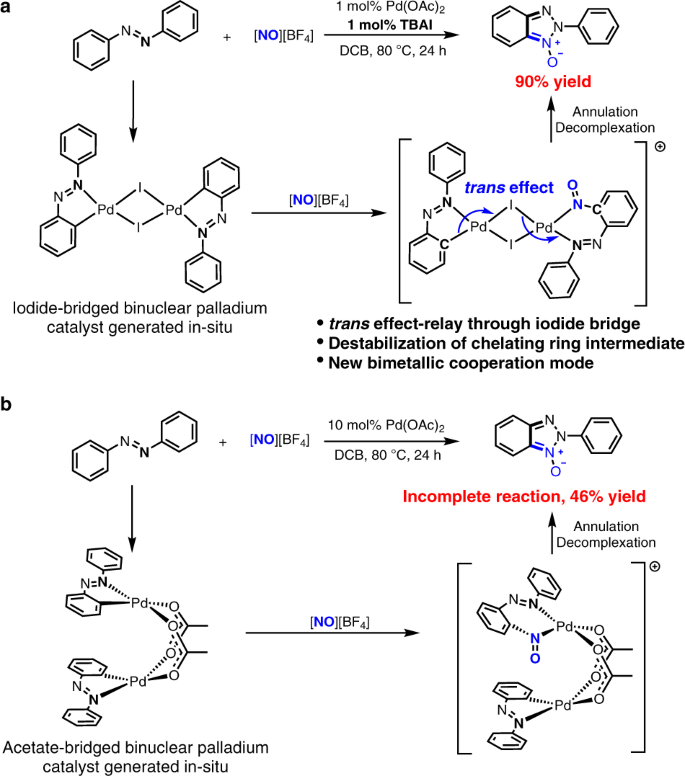
Iodide-enhanced palladium catalysis via formation of iodide-bridged binuclear palladium complex | Communications Chemistry